-
2009
Inception of company
-
2011
ISO 9001:2008 Quality Management System
-
2011
ISO 13485:2003 Quality Management System
-
2012
Interview in Economic Times
-
2012
CRISIL Healthcare Innovation Award for Transport Incubator and BILIPAD LED Phototherapy unit
-
2012
Economic Times’ Power of Ideas Award and cash prize
-
2013
CE Certificate for Infant Warmer, Phototherapy units, LED Phototherapy units, Transport Incubator
-
2013
Healthcare Innovation Award in Emergency Medical Service for Transport Incubator
-
2014
TOI Medicall Innovation Award for Transport Ventilator
-
2014
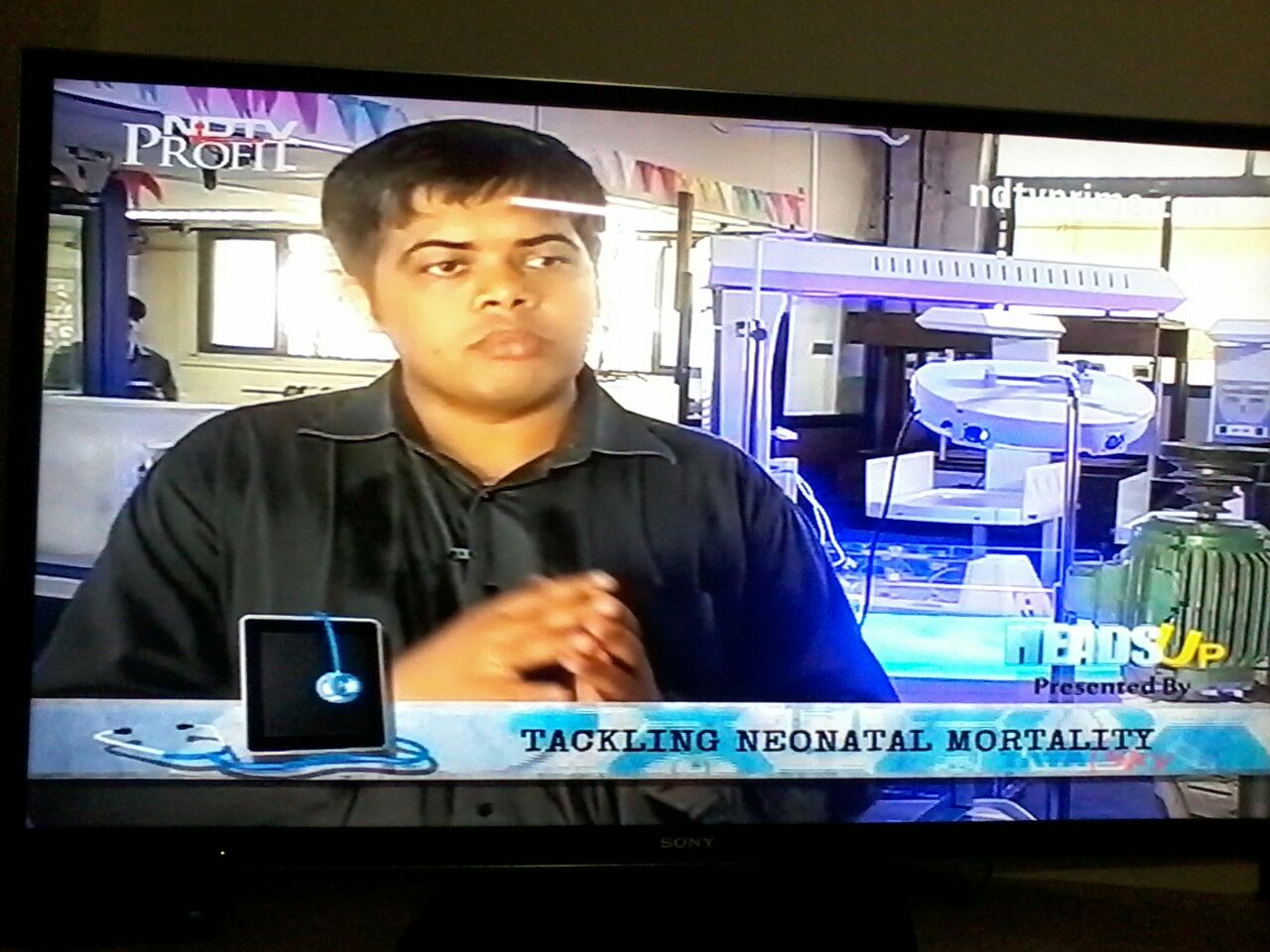
Interview by NDTV Prime
-
2015
Interview in Economic Times
-
2015
CE Certificate for Infant Warmer, Phototherapy units, LED Phototherapy units, Transport Incubator
-
2016
Honored at Rashtrapati Bhavan - Festival of Innovations - AVI Healthcare's Transport Incubator is amongst top 25 Innovative products in India
-
2016
Honored at Vigyan Bhavan
-
2017
ISO 13485:2003 / EN ISO 13485:2012 Quality Management System
-
2018
ISO 13485:2016 Quality Management System
-
2020
Expanded Factory to Vasai
-
2020
CE Certificate renewed for Infant Warmer, LED Phototherapy unit, Transport Incubator
-
2020
Tie up with IIT-K incubated company to manufacture Ventilators for Covid-19
-
2021
Launched our own Indigenous ICU Ventilators - VIHA dv10+ and VIHA dv10 Neonatal Ventilator
-
2021
Appreciated for efforts in COVID-19
-
2021
EN ISO 13485:2016 Quality Management System
-
2022
India Medical Device MSME of the Year - 2nd Runner Up
-
2022
Obtained Manufacturing License for Class A and B devices
-
2023
Obtained Manufacturing License for Class C devices
-
2025
First in India to obtain ICMED 13485 Plus sheme certificate
Who we are?
About Us – AVI Healthcare Pvt Ltd
AVI Healthcare Pvt Ltd, established in 2009, is a trusted name among medical device manufacturers in India, committed to delivering safe, reliable, and affordable healthcare solutions. We specialize in the design and manufacturing of life-saving technologies, including ICU ventilators and a comprehensive range of NICU equipment, tailored to meet the needs of hospitals, clinics, and neonatal intensive care units across the country and beyond.
With a strong focus on innovation and quality, AVI Healthcare has emerged as a leading ICU ventilators manufacturer, offering turbine-based ventilators designed for both adult and neonatal critical care. Our ventilators combine cutting-edge technology with user-friendly design, ensuring performance, portability, and versatility in even the most demanding clinical environments.
In addition to ventilators, we are one of the most reliable NICU equipment manufacturers, offering a broad portfolio of neonatal care products including:
-
Infant radiant warmers
-
LED phototherapy units
-
Bubble CPAP systems
-
Transport incubators
Our products are manufactured in India with a global outlook — ensuring compliance with international standards for quality, safety, and regulatory requirements.
Certified for Excellence and Global Compliance
At AVI Healthcare, quality and compliance are integral to our process. We operate a robust Quality Management System certified to EN ISO 13485:2016. Our manufacturing and documentation processes align with both EU MDR 2017/745and Indian Medical Device Rules (IMDR) 2017.
We take pride in being the first company in India to achieve the prestigious ICMED 13485 Plus certification, a nationally recognized scheme developed in alignment with global regulatory frameworks, and considered equivalent to the European CE marking. This distinction reinforces our commitment to safety, quality, and continuous improvement.
Furthermore, we manufacture and export CE certified medical devices, expanding our presence across Asia, the Middle East, and Africa. Our certifications demonstrate our ability to meet both domestic and international expectations for performance and compliance.
Our Mission: Making World-Class Healthcare Accessible
AVI Healthcare was founded with the vision of transforming healthcare access in India by delivering world-class medical technology at affordable costs. From product design to after-sales support, every step is driven by clinical needs, customer feedback, and technological advancements.
We proudly serve top government institutions, private hospitals, and healthcare providers across India. With installations spanning the country and growing exports, our goal is to make India a global hub for high-quality medical device manufacturing.
Why Choose AVI Healthcare?
-
Proven leader among medical device manufacturers in India
-
Certified ICU ventilators manufacturer with turbine-based technology
-
Wide range of neonatal products by a top NICU equipment manufacturer
-
Holders of ICMED 13485 Plus and CE certified medical devices
-
Commitment to affordable, reliable, and scalable healthcare solutions
At AVI Healthcare, it is our mission to become a “Glocal” (Global + Local) company manufacturing and Marketing Medical Equipment to treat, cure and save Premature and Newborn Infants by constant innovation, research and development, and achieving highest quality standards.
To be recognized across the length and breadth of the world as the leading company in providing affordable, safe and quality healthcare solutions.
We have made consistent investment in innovation, research and technology. We have been constantly innovating by customizing our products as per doctor’s needs and demands. We have the range of Neonatal products which are categorized as Neonatal Warming, Neonatal Transport, Neonatal Jaundice, Neonatal Resuscitation, Neonatal Ventilatory Support and Other Neonatal Products along with other medical products like ICU Ventilators
Click Here to know more about our products
Management Team

Mr. Laxmichand Gala
Mr. Laxmichand Gala, qualified BSc, founder of M/s Delta Medical Appliances in 1983, joined AVI Healthcare as mentor in 2011. He developed his brand DELTA in Surgical and Physiotherapy sector. In 1992, he got an opportunity to enter in the Neonatal field and since then there is no stop. With his perseverance, will and hard work he has developed cost-effective and quality products in the medical industry. He always believed in implementing quality management system to effectively produce quality products and accomplish various needs of Doctors, Government Hospital, Private Clinics, Individual etc. He has built a strong goodwill and has also recognized that after sales service forms an important factor for medical equipment. He has ensured that a strong distribution network, specially trained service engineers must be constantly on the toes to provide immediate 24 hours support on site, for quality and service go hand-in-hand. With this experience of over 35 years after nurturing DELTA, he has stepped up to nurture AVI Healthcare by his guidance, goodwill, wisdom and has been a pioneer of AVI Healthcare’s success.

Mr. Chirag Gala
Mr. Chirag Gala founded AVI Healthcare in 2009, with a vision to lower Infant Mortality by developing low-cost quality products. He is an innovator, with BE (Electronics) followed by Global MBA in Marketing from S.P. Jain Institute of Management and Studies. He always had a keen inclination towards research and development right from an early age. From developing a solar cooker in 8th grade to developing award winning and affordable products like Bilipad - LED Phototherapy unit and TransNANO Transport Incubator in 2012 TransVent Transport Ventilator in 2014; from selling foreign-manufactured Ventilators in 2009 to developing indigenous ICU Ventilators which is at par with international quality ventilators in 2021; he has shown the zeal to convert mere ideas and dreams into reality by hard work and passion. Chirag believes in developing and providing latest technologies at affordable prices. He insists that all AVI Healthcare products must meet national as well as international quality standards. Under his guidance, AVI Healthcare excels at par in achieving its motto in the healthcare sector.

Ms. Avni Gala
Ms. Avni Gala, a dual qualified Biomedical Engineer and Lawyer, joined AVI Healthcare in 2012. Leveraging her extensive expertise, she oversees quality assurance and regulatory compliance. She has been instrumental in establishing a robust quality framework for AVI Healthcare by developing comprehensive company protocols. Her dedication ensures all products and operations adhere to stringent national and international quality and regulatory standards.
Manufacturing Facility
We have about 17500 sq. ft. area (both sites combined) (Area: Site 1 – 2500 sq. ft. and Site 2 – 15000 sq. ft.) Site 1 has office setup and Research and Development (RnD) unit with latest technology. Site 2 includes entire manufacturing factory setup consisting of 6 floors. The factory unit follows EN ISO 13485 quality management system with dedicated and well-defined areas for Fabrication, Assembly, Stores, Quality and Testing, Packing and Dispatch with sufficient space for conducting all the activities.